Discover the seven business benefits of transparent product information management (PIM) in the pharmaceutical manufacturing industry
The business of pharmaceutical manufacturing is undergoing a data evolution. Business leaders need to understand how mastery of their data is essential to achieve leadership in a highly competitive market. The importance of the digital supply chain is growing. Manufacturers are building new processes that interconnect the back office with the front office and even directly to the customer’s operations. The pharmaceutical industry is becoming more digitally enabled and data intensive. Building a digital supply chain requires harvesting the raw materials that constitute it: Data.
.webp?width=581&name=pharmaceutical-manufacturing-product-information-management-2%20(1).webp)
Three challenges within the pharmaceutical manufacturing industry where data transparency can help:
-
Identifying new ways of delivering value to customers in the face of growing competitive pressure from lower cost alternatives
-
Ensuring patient safety by keeping up to date with evolutions in regulation and harmonization of industry codes of practice
-
Investing in digital transformation to build more cost-effective supply chains that address the changes in healthcare procurement and delivery models
The road to business transparency starts with data transparency
Your ability to confidently act upon key business data facts – such as product or customer data – across the enterprise requires data transparency. You achieve data transparency by implementing business-led processes that govern the ability to collect, enrich and share well-defined, accurate, coherent and up-to-date information across the enterprise and beyond.
Data transparency replaces ambiguity with actionable insight.
It’s difficult to make sure that your product information is transparent despite the clear business advantages that data transparency brings. Silos of organizations, applications and technologies all manage their own version of the truth. As a result, product information is fragmented. Furthermore, there is often no clear organization accountable for its construction, accuracy, coherence and provenance.
Siloed data management
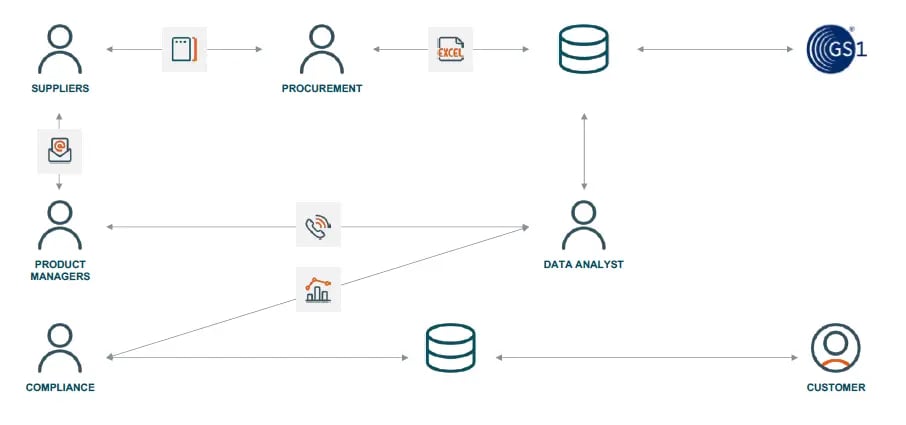
The problem of ambiguous data has its source in disparate data management. A siloed system architecture with no unified product information view - as exemplified above - creates a disarray of sources of information and accountability. The result is lack of transparency.
To achieve data transparency in pharmaceutical manufacturing, a unified product information management (PIM) lifecycle is needed to address the issues of siloed data (see illustration below).
A unified PIM solution must:
- Provide a business-led strategy for the collection, enrichment and dissemination of product information at an enterprise level
- Implement controls via an organization, as well as processes and tooling to ensure the data that describes products is fully transparent and fit for purpose
- Work alongside (rather than replace) existing infrastructure, applications and processes in order to achieve rapid business benefits with limited impact to existing operations
Unified product information management
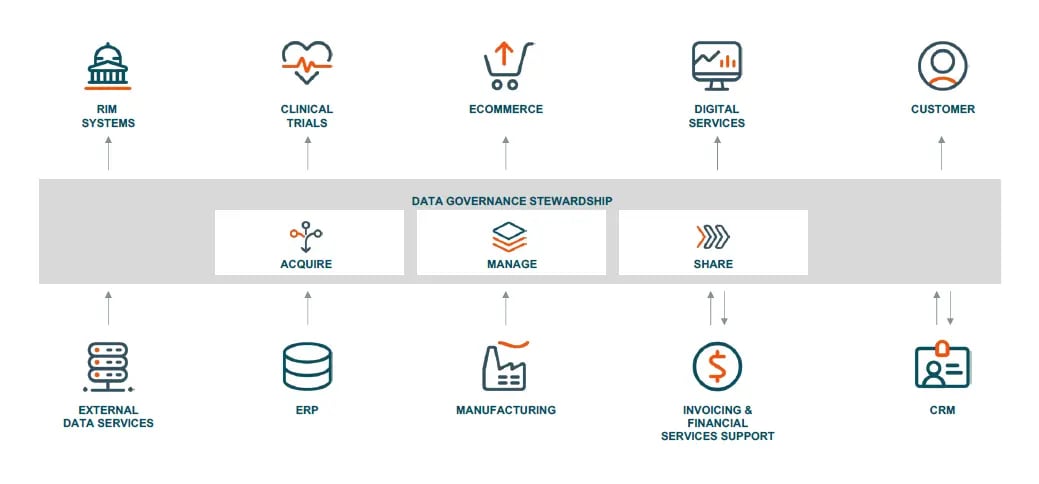
7 ways in which a unified product information management lifecycle can help in the pharmaceutical manufacturing business
1. Improve patient safety
Having access to clear and complete product information plays an important part in the prevention of adverse events linked to information mishandling. In case of adverse events, regulators require that their agencies be informed via centralized reporting systems in electronic format to ensure prompt and appropriate response.
A PIM solution can help by: Providing centralized, business-led controls on the completeness, accuracy and availability of product information.
2. Reduce costs of data errors in regulatory requirements
As the healthcare industry continuously evolves to serve patients better, codes for industry ethics strive for greater transparency. Regulatory information management (RIM) systems need harmonized and coherent product information in order to establish the basis for reporting using, for example, CTD or eCTD for pharmaceuticals for human usage.
Declarations to local and regional authorities require that information abides by specific organizational rules and structures. These need to be established in a unified way to ensure coherence and completeness. The Sunshine Act and EFPIA code are examples of how spending reporting requires not only good HCO/HCP information, but also reliable product information.
A PIM solution can help by: Providing business-led controls on the product data that feeds compliance functions, thus avoiding costs and penalties associated with data errors.
3. Increase business transparency with better product traceability
Manufacturers are adopting global standardization efforts such as IDMP to improve product traceability and patient safety. The IDMP system uses standardized product description formats developed by the ISO that cover substance, product, organization and referential (SPOR) data.
The standards provide a means of exchanging and tracing information between various entities within the ecosystem, such as different companies and between regulators and marketing authorization holders (MAHs). But they can also be seen as an effective means of exchanging information between different departments within an organization. IDMP includes the use of GS1 GTINs to facilitate the communication between MAHs, the supply chain, business partners and traceability functions.
A PIM solution can help by: Ensuring that product and packaging descriptions conform to standards for identification and traceability.
4. Help your customers to find and order your products more easily
Aligning on normalized product coding can facilitate the ordering process for your customers. Healthcare organizations (HCOs) and group purchasing organizations (GPOs) are implementing automated e-procurement processes in order to reduce cost and increase efficiency.
Key to success is having normalized product data, often obtained from data pools, to eliminate ordering errors. Some HCOs are making it mandatory for their suppliers to use GS1. Some buying organizations impose penalties for incorrect information that delays their procurement operations.
A PIM solution can help by: Synchronizing accurate product information with GS1-certified data pools and customer e-procurement portals.
5. Educate your employees and your customers
Learning management systems (LMS) need in-depth and accurate product reference data to provide the most up-to-date information in the best format possible to better serve internal employees, distributors, customers and patients.
A PIM solution can help by: Organizing and making available the product information, including identification, description, packaging, regional variations, imagery, digital assets, MSDS, etc.
6. Make better, data-driven decisions
Being able to take confident and immediate action on the data you receive can change business outcome. With transparent product data you can gain more insight into your business, such as understanding which products perform best via which sales channels, and anticipate rather than react to demand.
A PIM solution can help by: Organizing products into categories and hierarchies that support better analytics for business insight.
7. Build efficiency with digital supply chains
Normalizing product descriptions across the organization and into the customer’s hands can help support the journey to automate more internal processes and digitize operations. Supplier catalog on-boarding, raw materials management, e-procurement, e-sales, remote inventory management and replenishment, fulfillment and many other processes are targets for joined-up automation enabled with a unified and accurate PIM process. Having a highly granular level of product information enables companies to drill into product lines and understand how and where specific medical devices were manufactured and which third parties supplied components and services.
A PIM solution can help by: Providing product information in the format and context needed for consumption by all systems and applications within the supply chain.
EXPLORE
Master Data Management for Life Sciences